Huntington's disease drug: Teva Pharma settles patent dispute with Lupin, continues to litigate against Aurobindo
PDR0000401 An electronic inhaler add-on module which store and transmit information related to inhaler use (eModule) Teardown Internal Photos Teva Branded Pharmaceutical Products R&D;, .
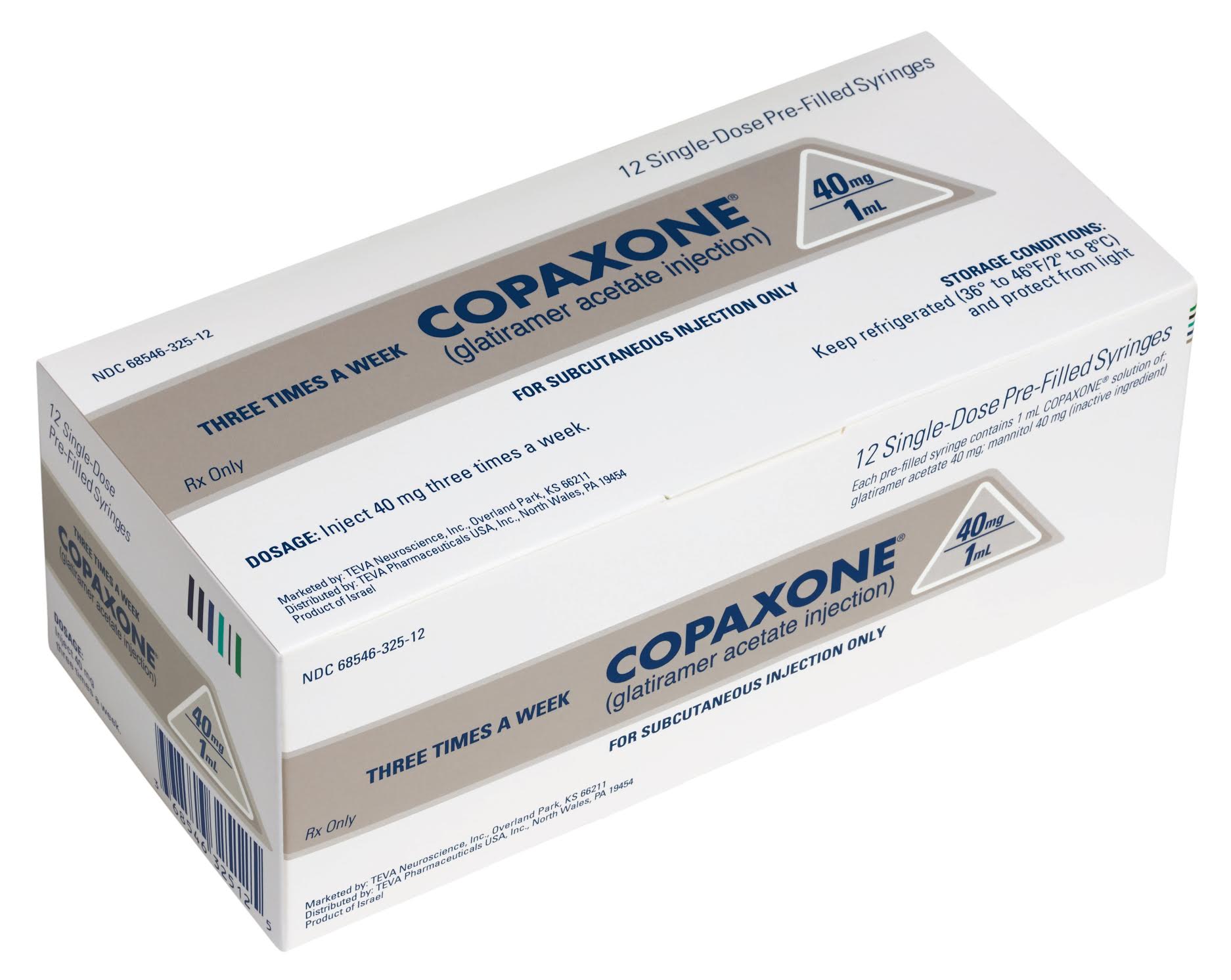
Uninhibited' price hikes, lobbying and more: Congressional probe gives detailed look into Teva's long run with Copaxone | Fierce Pharma